What is HFpEF?
Heart failure with preserved ejection fraction (HFpEF) is a heterogeneous syndrome, frequently associated with high rates of hospitalization and mortality. However, there is still a great deal to learn about the condition, which affects more than 50% of the 64 million cases of heart failure worldwide. Its increasing prevalence, paired with the challenges of its diagnosis and therapy, means that many patients are left vulnerable to a high risk of mortality. 1
Guide To HFpEF
Learn more about the existing knowledge on the etiology and differences between HFpEF and HFmrEF, and discuss the issues around diagnosing HFpEF, according to guidelines.
What is the definition of HFpEF in medical terms?
HFpEF - Heart failure with reduced ejection fraction
Heart failure with reduced ejection fraction (Heart failure with preserved ejection fraction (HFpEF) is a clinical syndrome in which patients have signs and symptoms of heart failure as the result of high left ventricular (LV) filling pressure, despite normal or near-normal LV ejection fraction (LVEF; ≥50 percent).


HFrEF
When the heart can’t pump blood effectively, its left ventricular (LV) function is impaired. This leads to a reduction in its ejection fraction (EF), which is the volume of blood ejected from the left ventricle at each heartbeat.
An ejection fraction of less than 40% is classified as heart failure with reduced ejection fraction (HFrEF).
HFpEF
With HFpEF, heart failure is occurring despite ejection fraction remaining within normal ranges. This happens because the heart muscle has become so thick and stiff that the ventricle is no longer able to relax properly and holds a smaller-than-usual volume of blood. When the LV pumps out this smaller amount of blood seemingly effectively, the EF measurement falsely shows a normal percentage of blood being pumped. However, the reduced amount of blood the LV pumps isn’t enough to meet the body’s needs. [2]



|
Related read: HFpEF and HFrEF are complex conditions often requiring different modes of detection and therapy. Our article, HFpEF vs HFrEF, goes into greater detail about both the similarities and differences between them. It covers the structural, molecular, and cellular mechanisms that contribute to HFpEF and HFrEF, and the other key differences in their risk factors, diagnosis, and treatment. |
Causes of HFpEF
As HFpEF is a complex, multisystem loss of cardiac and vascular reserve capacity, it can be challenging to pinpoint exactly what causes HFpEF. A multitude of studies have attempted to uncover the cellular and molecular basis of the condition, but we still don’t have a full understanding of its pathophysiology. Additionally, contributing factors can vary from patient to patient. HFpEF is usually the result of a multitude of reserve impairments that combine to cause symptomatic heart failure.
Still, some connections have been made between the onset of HFpEF and chronic inflammation resulting from conditions such as diabetes, hypertension, obesity, and a sedentary lifestyle. However, other comorbidities also come into play in the pathology of the disease. These make it challenging to pinpoint the exact causes of HFpEF in any given patient.
HFpEF comes with a high risk of comorbidities
HFpEF is often associated with comorbidities that lead to chronic inflammation, cardiomyocyte hypertrophy, and fibrosis, which all lead to structural and cellular alteration of the heart. These eventually lead to diastolic dysfunction and an inability of the left ventricle to relax properly. [3]
HFpEF is frequently associated with comorbidities such as:
- hypertension (60%-80%)
- ischemic heart disease (35%-70%)
- diabetes (20%-45%), and
- atrial fibrillation (15%-40%)
A distinct pathophysiology
HFpEF also presents with differing pathophysiologies to HFrEF. While HFpEF tends to be associated with structural changes brought on by chronic inflammation, the structural alterations in HFrEF are usually brought on by factors such as acute inflammation (for example, from a virus), trauma to the heart, and substantial cardiomyocyte loss. This leads to the development of systolic dysfunction, or the ability of the left ventricle to contract properly.
Causes of HFpEF vary, and in many cases can be unclear. To get a more in-depth look at HFpEF’s pathophysiology, including why females tend to present with HFpEF more often, read our article about what causes heart failure with preserved ejection fraction. This detailed article also covers how cardiac aging can play a role in HFpEF onset, and the clinical indicators that can chart its pathophysiology.

HFpEF diagnosis
Issues around the detection of HFpEF
HFpEF presents a significant challenge to the diagnostic process as it lacks a useful and objective one-method-fits-all approach.
HFpEF may be represented by different pathophysiological phenotypes, which require differential identification and management. Current diagnostic algorithms include clinical, laboratory, and instrumental characteristics, sophisticated imaging modalities, and invasive measurements.
Many of these factors can be unclear, which leads to diagnosis by exclusion, inconclusive diagnosis, or a missed diagnosis altogether.
The “gold standard” for HFpEF diagnosis, according to the 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure, is considered:
- The presence of signs and symptoms of HF;
- A preserved left ventricular (LV) EF
- An elevated LV diastolic pressure at rest or exercise by cardiac catheterization. [7]
Two-dimensional and Doppler echocardiography has been used to noninvasively estimate LV filling pressure and incorporated into diagnostic algorithms for HFpEF in guidelines. However, it suffers from many variables. Additionally, some patients' LV filling pressures are only elevated at invasive stress testing, but are neither universally available nor desirable.
The sensitivity, however, varies substantially for this approach, ranging from 25% to 80%. In addition, patients at the early stage of disease may be assessed as normal or indeterminant.[8]
Inconclusive
Clinical and echo variables
Invasive
|
Related read: Our article, Diagnosing Heart Failure with Preserved Ejection Fraction, goes into greater detail about how HFpEF is diagnosed according to current clinical guidelines; including history and physical examination, imaging via rest and stress, natriuretic peptides, staging, and new integrated approaches. |
How is artificial intelligence supporting HFpEF diagnosis?
Radical progress is being made
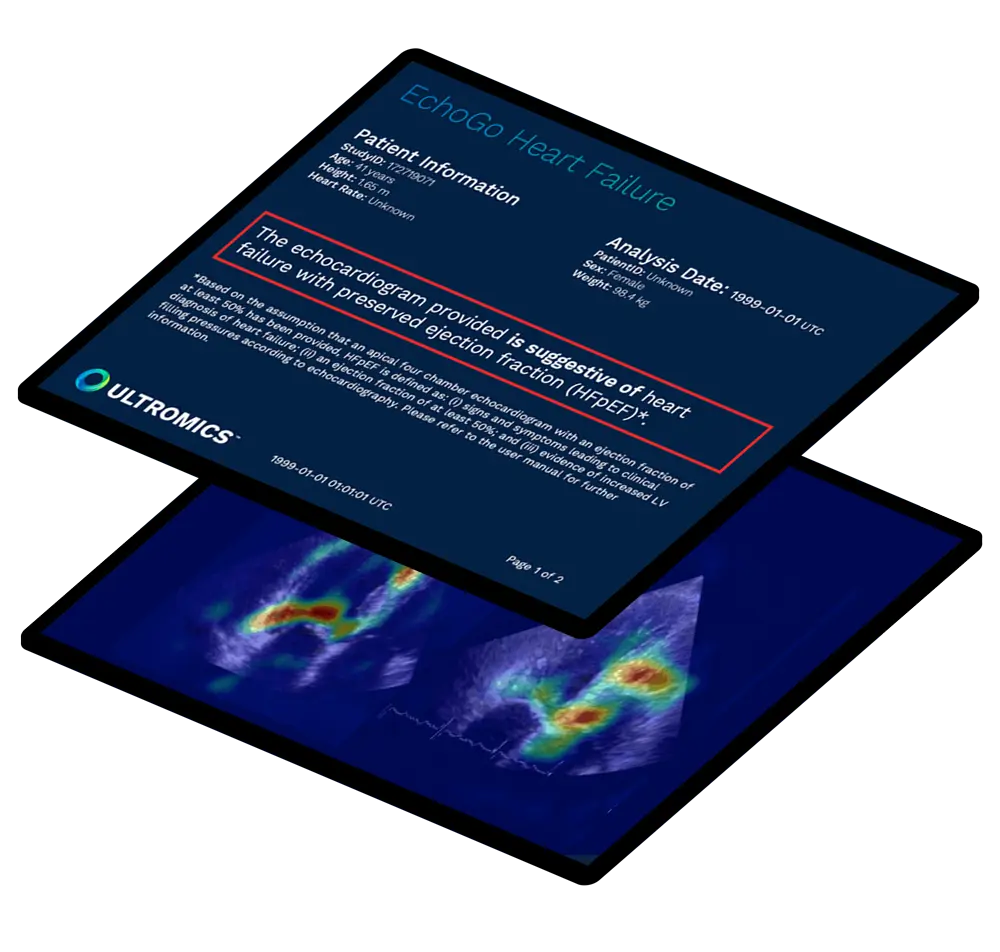
There is an urgent need to develop precise tools to help diagnose HFpEF patients so they can receive faster access to life-saving new treatment. EchoGo Heart Failure, from Oxford University and built in collaboration with the Mayo Clinic, delivers precision HFpEF detection using the power of AI from a single apical 4-chamber view of an echocardiogram.
The AI in EchoGo Heart Failure can be applied for HFpEF and marks a huge leap forward from traditional diagnostic algorithms for HFpEF.
74%
of cases correctly reclassified that were otherwise indeterminate.

How can HFpEF be treated?
Treatment
To date, the exact mechanisms and effective treatment strategies for HFpEF are still poorly understood. Therapy has largely been focused on the management of symptoms and comorbidities, such as medications to lower blood sugar in diabetic patients. Evidence published in the EMPOROR-PRESERVED trial shows sodium-glucose cotransporter 2 inhibitors (SGLT2is) are the first drug class to improve cardiovascular outcomes in HFpEF. [10] Recommended in the 2022 HF guidelines, SGLT2is have demonstrated a relative risk reduction in heart failure hospitalizations by 29%. [11]
29% reduced risk of hospitalization
In addition to results published in the EMPOROR-PRESERVED trial. The DELIVER trial for another SGLT2i Dapagliflozin showed a reduction in worsening heart failure which included any of:
- An unplanned hospitalization for heart failure
- An urgent visit for heart failure
- Cardiovascular death
Results found Dapagliflozin reduced the rates of these end points versus the placebo.
16.4% Dapagliflozin
19.5% Placebo

The double impact of an efficient and precise HFpEF diagnosis
Improved patient impact
HFpEF represents a growing epidemic for patients. The symptoms, which include shortness of breath, chest discomfort, and debilitating fatigue, can have an enormous impact on a person’s quality of life. It is often deadly, carrying a five-year survival rate of just 35% to 40%.
Receiving an efficient and precise HFpEF diagnosis can provide faster access to treatment, significantly improving a patient’s quality of life.
Poor survival rate
High hospital admissions
Over 50% rehospitalization
$70 billion annual spend
Envisioning a better outlook for HFpEF patients
Partners supporting HFpEF detection
The National Institutes of Health (NIH) and the National Heart Lung and Blood Institute (NHLBI), managed by the Foundation for the National Institutes of Health (FNIH), have come together with key partners such as Ultromics to form The Accelerating Medicines Partnership® Heart Failure (AMP® HF).
The 5-year, $37 million multi-stakeholder collaboration brings together partners from both public and private sectors across the world to investigate HFpEF.
This partnership comes in response to the overwhelming need to better understand HFpEF.


Frequently asked questions (FAQs)



You can find more information about complete and ongoing trials with AI for HFpEF on our Clinical Trials Proof page.
The US ICD claim code for HFpEF is I50.3.
Code I50.3 is applicable to:
- Diastolic left ventricular (LV) heart failure
- Heart failure with normal ejection fraction
- Heart failure with preserved ejection fraction (HFpEF)
The ICD code I50.3 applies to HFpEF in general, which means it should not be used for reimbursement purposes. Instead, its sub-codes should be used, as these contain a greater level of detail.
These codes are:
- I50.30: Unspecified diastolic (congestive) heart failure
- I50.31: Acute diastolic (congestive) heart failure
- I50.32: Chronic diastolic (congestive) heart failure
- I50.33: Acute on chronic diastolic (congestive) heart failure [2]
For more information about ICD claim codes for HFpEF, including tips for best practices while reporting, such as specifying acuity and type,
read our article: What is the ICD Code for HFPEF?
Simply fill in this form here for a free demo showing you how our platform can help improve the detection of HFpEF.
References:
- Urbich M, Globe G, Pantiri K, et al. A Systematic Review of Medical Costs Associated with Heart Failure in the USA (2014–2020). PharmacoEconomics. 2020:19;38(11):1219–36.
- Borlaug BA, Paulus WJ. Heart failure with preserved ejection fraction: pathophysiology, diagnosis, and treatment. European Heart Journal. 2010 Dec 7;32(6):670–9.
- Simmonds SJ, Cuijpers I, Heymans S, Jones EAV. Cellular and Molecular Differences between HFpEF and HFrEF: A Step Ahead in an Improved Pathological Understanding. Cells. 2020 18;9(1):242.
- Sanders‐van Wijk S, Barandiarán Aizpurua A, Brunner‐La Rocca H, et al. The HFA‐PEFF and H2FPEF scores largely disagree in classifying patients with suspected heart failure with preserved ejection fraction. European Journal of Heart Failure. 2020 2;23(5):838–40.
- Cardoso R, Graffunder FP, Ternes CMP, et al. SGLT2 inhibitors decrease cardiovascular death and heart failure hospitalizations in patients with heart failure: A systematic review and meta-analysis. EClinicalMedicine. 2021 ;36(5):100933.
- Nanayakkara S, Patel HC, Kaye DM. Hospitalisation in Patients With Heart Failure With Preserved Ejection Fraction. Clinical Medicine Insights: Cardiology [Internet]. 2018;12:117954681775160. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5764137/
- Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA guideline for the management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation [Internet]. 2022 1;145(18). Available from: https://www.ahajournals.org/doi/10.1161/CIR.0000000000001063
- Paulus WJ. H2FPEF Score. Circulation. 2018 28;138(9):871–3.
- Egashira K, Sueta D, Komorita T, et al. HFA-PEFF scores: prognostic value in heart failure with preserved left ventricular ejection fraction. The Korean Journal of Internal Medicine [Internet]. 2022;37(1):96–108. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8747922/#:~:text=Calculation%20of%20the%20HFA%2DPEFF%20score&text=A%20total%20score%20
- Kumbhani DJ. Empagliflozin Outcome Trial in Patients With Chronic Heart Failure With Preserved Ejection Fraction [Internet]. American College of Cardiology. 2023. Available from: https://www.acc.org/latest-in-cardiology/clinical-trials/2021/08/25/23/07/emperor-preserved#:~:text=Highlighted%20text%20has%20been%20updated
- AJMC. Key Takeaways from the 2022 ACC/AHA/HFSA Guideline for the Management of Heart Failure. Supplements and Featured Publications [Internet]. 2022:29; Available from: https://www.ajmc.com/view/key-takeaways-from-the-2022-acc-aha-hfsa-guideline-for-the-management-of-heart-failure
- Boehringer Ingelheim. Unblur the world of HFpEF [Internet]. Boehringer-ingelheim.com. 2023. Available from: https://pro.boehringer-ingelheim.com/us/focusonhfpef/
Curious about upcoming research and innovation?
Sign up to hear about the latest news.